When you buy through our links, we may earn a commission. Products or services may be offered by an affiliated entity. Learn more.
Sleep Apnea Treatment
- Sleep apnea is a disorder characterized by interrupted breathing during sleep.
- Depending on the severity of sleep apnea, treatment options include PAP therapy, oral appliances, surgery, and lifestyle changes.
- CPAP is typically the first treatment recommended, but a sleep specialist may prescribe an alternative pressurized air device depending on your situation.
- Consult a healthcare provider for a proper diagnosis and personalized treatment plan.
There are two main types of sleep apnea, both of which require treatment. Obstructive sleep apnea (OSA) involves pauses or reduced breathing during sleep due to complete or partial airway blockages. By contrast, for people with central sleep apnea (CSA), breathing stops during sleep as a result of issues with the brain or nervous system.
It is important to properly diagnose and treat sleep apnea. People with OSA face an increased risk for high blood pressure, type 2 diabetes, and other health issues. Sleep apnea treatment can help reduce the risk of these complications.
Not Sure if You Have Sleep Apnea? Try an At-Home Test

our partner at sleepdoctor.com
Save 54% on your Sleep Test Today
Shop now“Truly grateful for this home sleep test. Fair pricing and improved my sleep!”
Dawn G. – Verified Tester
Therapy Treatments for Sleep Apnea
Multiple non-invasive treatment options for sleep apnea exist, some of which are more effective than others. Non-surgical options fall into two categories: positive airway pressure (PAP) devices and oral appliances.
Continuous Positive Airway Pressure (CPAP)
Doctors consider continuous positive airway pressure (CPAP) the gold standard treatment for OSA, and it is usually the first treatment offered to people diagnosed with moderate or severe obstructive sleep apnea. Generally, a CPAP machine plugs into an outlet and sits next to a sleeper’s bed. A tube connects the machine to a mask that covers the sleeper’s mouth, nose, or both. Then, the CPAP machine blows air into the sleeper’s airway, which helps keep it open during sleep.
CPAP is considered the most effective OSA treatment, but it must be used nightly. CPAP therapy is less effective at treating central sleep apnea.
CPAP machines only push out air at one rate, which is calibrated by the doctor to the average rate needed by the sleeper. Since some people have trouble tolerating this continuous level of air pressure, other options are available.
Bilevel Positive Airway Pressure (BiPAP)
Sometimes doctors prescribe a bilevel positive airway pressure (BiPAP or BPAP) machine when a person with sleep apnea cannot tolerate CPAP. The BiPAP machine operates similarly to a CPAP machine, with a connected tube that pushes air into a mask to keep the airway open. However, a BiPAP machine differs from a CPAP machine in that it releases air at a higher pressure during a sleeper’s inhale and at a lower pressure during a sleeper’s exhale.
This feature makes BiPAP therapy a good choice for sleepers who have trouble exhaling into the stream of higher-pressure air coming from a CPAP machine. BiPAP machines are also sometimes prescribed to people who have sleep apnea as well as severe obesity or certain other health conditions, like chronic obstructive pulmonary disease and hypoventilation.
Auto-Adjusting Positive Airway Pressure (APAP)
Auto-adjusting positive airway pressure machines, often called APAP or auto-CPAP machines, also work similarly to CPAP machines. APAP machines differ from CPAP machines with their ability to automatically adjust the pressure of air they release. This ability can make the machine more comfortable to use than a CPAP device, since sleepers need different levels of pressure at different times of night, depending on their sleeping position or other factors.
APAP machines use pressure sensors and a computer algorithm to auto-titrate, or calculate exactly what pressure of air a sleeper needs at any given moment. As a result, an APAP machine might work better for someone who feels uncomfortable with the consistent pressure of air released from a CPAP machine. Sleep specialists can program APAP machines to only release air at pressures that fall within a preset minimum and maximum, to help ensure its effectiveness.
Adaptive Servo-Ventilation (ASV)
Adaptive servo-ventilation (ASV) is used primarily as treatment for people who experience central sleep apnea. ASV operates similarly to PAP therapy, delivering pressurized air through a tube and mask that the sleeper wears. However, rather than delivering a set level of air on inhalation and exhalation, ASV is programmed to release customized air pressure that adapts in real time, anticipating and responding to central apneic events.
Initially, ASV was used to treat Cheyne-Stokes respiration, a specific pattern of breathing that sometimes occurs due to heart failure . While adaptive servo-ventilation is useful for people experiencing central sleep apnea under certain conditions, studies have shown that ASV may be potentially harmful for people with both CSA and some types of advanced congestive heart failure .
Expiratory Positive Airway Pressure (EPAP)
Unlike CPAP and BiPAP sleep apnea treatments, an expiratory positive airway pressure (EPAP) device is not a motorized machine. Rather, EPAP treatment consists of two small valves that fit inside the nostrils. EPAP therapy provides airway pressure by creating resistance that keeps the airway expanded when a person exhales.
EPAP therapy can be more convenient for some sleepers, since it does not require electricity, creates little to no noise, and is small and lightweight. Nasal EPAP therapy has generally been evaluated for use in people with OSA, not CSA.
EPAP therapy is newer and it is less commonly prescribed than the other PAP therapies. However, a review of EPAP studies found that people using EPAP experience a 53% reduction in OSA symptoms.
Some researchers recommend using EPAP for people with mild to moderate OSA who cannot tolerate CPAP therapy. Other studies suggest EPAP works well for people with severe OSA , as well. A more recent study found that combining EPAP with an oral device called a mandibular advancement splint effectively treats OSA for people who have trouble reducing sleep apnea symptoms with just the oral device.
Oral Appliances
Oral appliances are designed to reduce OSA symptoms by physically opening the airway. Like EPAP, oral appliances are often only prescribed after a person has found that they cannot tolerate one of the PAP machine treatments. Two oral appliances are most common.
- Mandibular advanced splints (MAS): This treatment involves wearing a custom-made dental device that fits over the top and bottom teeth, keeping the lower jaw pulled forward. By keeping the jaw forward, MAS also keeps the tongue forward, increasing the chance the airway will stay open. MAS may be best utilized in cases of disruptive snoring and mild to moderate OSA.
- Tongue-retaining devices: These devices are meant to keep the airway clear by keeping the tongue positioned forward, using suction. Tongue-retaining devices have been demonstrated to help reduce OSA symptom severity, though they are not as effective as CPAP. Researchers suggest they might be better for short-term use.
Take Our Quiz to Know Your Sleep Apnea Risk
If you’re still not sure if you have sleep apnea, we encourage you to take our short quiz below to understand whether you may be affected.
Surgical Treatments for Sleep Apnea
When non-invasive devices fail to adequately treat sleep apnea, a sleep specialist may recommend surgery to help prevent lapses in breathing during sleep. The type of surgery a person might undergo depends on what is causing their breathing issues.
Tissue Removal or Shrinkage
Several tissue removal or shrinkage surgeries may help to treat OSA caused by identifiable blockages:
- Radiofrequency tissue reduction: Using a newer surgical technique called radiofrequency surgery , surgeons remove tissue blocking the airway with a high-frequency current. This type of surgery can be used to stiffen and reduce the size of the soft palate. It can also be used to remove tissue from the nose, tonsils, or tongue base.
- Uvulopalatopharyngoplasty: This type of surgery involves removing part of the uvula, the bell-shaped tissue dangling at the back of the mouth, along with parts of the soft palate. Often, this type of surgery is laser-assisted, but more recent research has found that laser-assisted uvulopalatoplasty may worsen apnea symptoms in some people.
- Adenoidectomy: Doctors may also be able to help clear the airway by removing the adenoids, glands found above the roof of the mouth. This surgery is most common in children with OSA, and it often occurs at the same time as tonsil removal.
- Tonsillectomy: Removing the tonsils is a common OSA surgical treatment that may be done along with other tissue-removing surgeries.
- Tracheotomy: A tracheotomy involves creating a hole in the neck, so air can enter directly into the windpipe through a tube. This is the most effective OSA surgery, but it is also the most invasive. Experts recommend only using tracheotomy when a person’s life is at risk and all other options have been exhausted.
Jaw Repositioning
Maxillomandibular advancement involves permanently moving the jaw forward to help keep the airway clear. Studies show that this surgery reduces sleep apnea symptoms by 87% on average and is successful in 85% of people. However, it is comparatively invasive. Maxillomandibular advancement comes with more risks and can impact a person’s appearance.
Tongue-Related Surgeries
Three tongue-related surgeries are sometimes conducted to alleviate OSA symptoms:
- Genioglossus advancement: This surgery focuses on the genioglossus muscle , which makes up most of the visible tongue. By surgically advancing the tongue forward in the mouth, it is less likely to fall back and block the airway.
- Hyoid suspension: The hyoid bone sits toward the top of the neck and is sometimes called the tongue bone, because of its role in holding the tongue in place. Hyoid suspension surgery is intended to alter tongue placement to open up the airway. It is often combined with other surgical procedures.
- Midline glossectomy: This surgery involves removing part of the tongue in order to reduce the space it takes up and clear the airway. Studies show this surgery can be successful for reducing OSA symptoms, especially when paired with other surgical procedures.
Nasal Surgeries
In some cases, the structure of a person’s nose and nasal passage contributes to OSA symptoms. For example, a deviated septum, nasal growths, and excessively large bones within the nose can all cause or exacerbate OSA. Many nasal issues can be treated through traditional surgery or minimally invasive radiofrequency surgery. Studies of such surgeries have not returned high success rates, but nasal surgery may improve outcomes for other types of OSA treatments.
Nasal surgeries sometimes performed for OSA include:
- Rhinoplasty, which changes the shape of the nose
- Septoplasty, which corrects a deviated septum
- Endoscopic sinus surgery, which helps clear the sinuses
- Nasal valve surgery, which opens up the nostrils or the internal nasal valves
- Turbinectomy, which removes or shrinks some of the tiny bones in the nose
Nerve Stimulation
Two nerve stimulation surgeries are available for people with sleep apnea. Hypoglossal nerve stimulation treats OSA, while transvenous phrenic nerve stimulation treats CSA.
- Hypoglossal nerve stimulation: The hypoglossal nerve connects to the tongue. Electrically stimulating this nerve can cause the tongue to push forward, clearing the airway. In this outpatient surgery, an implant is put into the chest along with a sensor to monitor breathing and a stimulator connected to the hypoglossal nerve. Also referred to as upper airway stimulation, hypoglossal nerve stimulation is a newer treatment recommended for people who have moderate to severe OSA and cannot tolerate a PAP machine.
- Transvenous phrenic nerve stimulation: The phrenic nerve is closely involved in breathing, as it connects with the lungs and diaphragm. Electrically stimulating this nerve using a surgical implant has shown promising results as a CSA treatment.
Lifestyle Changes to Treat Sleep Apnea
Multiple lifestyle factors may be able to reduce severity of OSA symptoms.
- Weight loss: Research shows that losing excess weight can lead to an improvement in OSA symptoms. For this reason, medical professionals might recommend diet and exercise for weight management.
- Throat exercises: Also called oropharyngeal exercises, these repeated movements of the tongue, soft palate, and throat have been demonstrated to reduce OSA symptom severity. In several studies using these exercises, participants saw improvements in OSA symptoms after doing the exercises regularly for three months.
- Changing sleep position: Positional therapy can reduce OSA symptom severity in some people. Back sleeping is associated with more severe symptoms for many people with OSA, so positional therapy generally involves trying to train a person to stop sleeping on their back.
- Avoiding alcohol and certain medications: People with OSA should avoid alcohol and sedatives in the hours before sleeping, if possible. Alcohol is thought to increase OSA symptoms by relaxing muscles associated with the airway, thereby causing blockages.
- Quitting smoking: Smoking cigarettes increases a person’s risk of having OSA, so doctors often recommend that smokers with OSA quit smoking. However, more research is needed to understand the relationship between smoking and OSA.
Recommended Products for Sleep Apnea
From Our Partners at SleepDoctor.com
How to Pick The Right Sleep Apnea Treatment For You
Usually, a person’s sleep specialist determines which sleep apnea treatment is best for them based on their unique symptoms and health situation. Along with treatment of any underlying health conditions, a CPAP machine is usually the first treatment prescribed for sleep apnea. CPAP therapy is considered most effective and reliable in those who are able to adhere to treatment. If a sleeper has trouble tolerating CPAP therapy, their sleep specialist may next prescribe BiPAP, ASV, EPAP, or an oral device as an alternative treatment. Surgery is often only considered after non-invasive treatments have been tried.
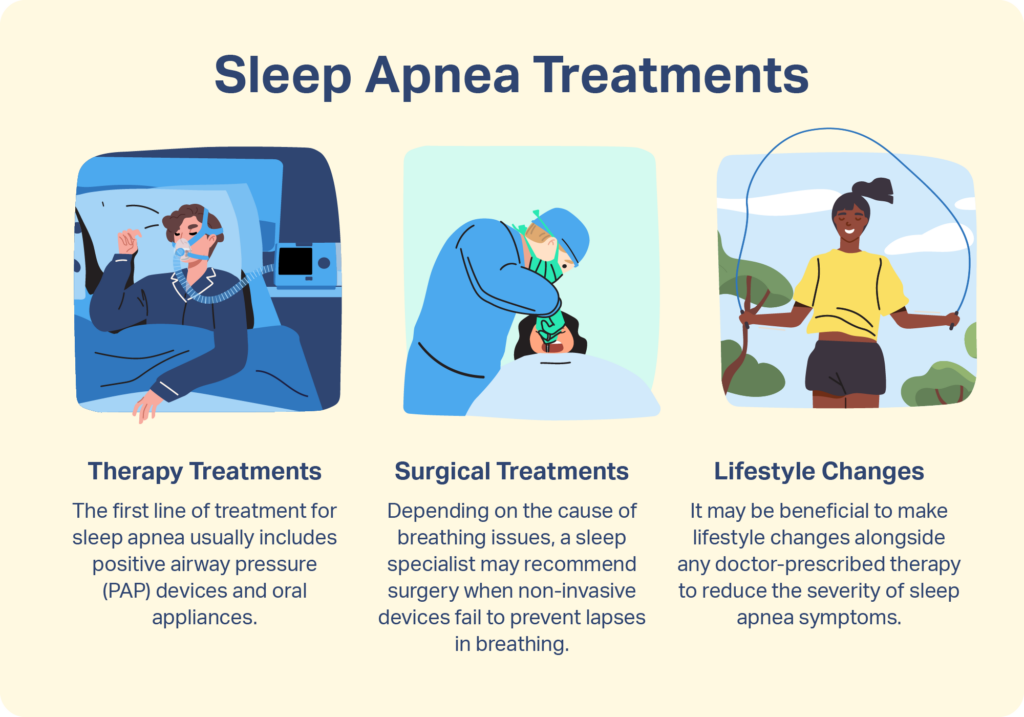
Many treatments for sleep apnea require ongoing use of a device. If you have sleep apnea, you may try making lifestyle changes alongside any therapy your doctor prescribes. However, you should also continue to use treatments prescribed by your doctor or sleep specialist until directed otherwise.
Talking With Your Doctor About Sleep Apnea Treatment Options
Once your sleep specialist gives you a sleep apnea diagnosis, they will outline their plan for your treatment. If you have trouble adhering to a treatment, such as CPAP therapy, make another appointment with your doctor or sleep specialist to see if it is appropriate to stop the treatment and to find out what other treatments they recommend trying next.
Medical Disclaimer: The content on this page should not be taken as medical advice or used as a recommendation for any specific treatment or medication. Always consult your doctor before taking a new medication or changing your current treatment.
References
13 Sources
-
American Academy of Sleep Medicine. (2014). The International Classification of Sleep Disorders – Third Edition (ICSD-3). Darien, IL.
https://aasm.org/ -
Jaffuel, D., Philippe, C., Rabec, C., Mallet, J. P., Georges, M., Redolfi, S., Palot, A., Suehs, C. M., Nogue, E., Molinari, N., & Bourdin, A. (2019). What is the remaining status of adaptive servo-ventilation? The results of a real-life multicenter study (OTRLASV-study): Adaptive servo-ventilation in real-life conditions. Respiratory Research, 20(1), 235.
https://pubmed.ncbi.nlm.nih.gov/31665026/ -
Patel, A., Perez, I., & Rabiei-Samani, S. (2021). What is adaptive servo-ventilation (ASV)? American Journal of Respiratory and Critical Care Medicine, 204(2), P3–P4.
https://pubmed.ncbi.nlm.nih.gov/34283697/ -
Lorenzi-Filho, G., Almeida, F. R., & Strollo, P. J. (2017). Treating OSA: Current and emerging therapies beyond CPAP. Respirology (Carlton, Vic.), 22(8), 1500–1507.
https://pubmed.ncbi.nlm.nih.gov/28901030/ -
Wu, H., Yuan, X., Zhan, X., Li, L., & Wei, Y. (2015). A review of EPAP nasal device therapy for obstructive sleep apnea syndrome. Sleep & Breathing, 19(3), 769–774.
https://pubmed.ncbi.nlm.nih.gov/25245174/ -
Berry, R. B., Kryger, M. H., & Massie, C. A. (2011). A novel nasal expiratory positive airway pressure (EPAP) device for the treatment of obstructive sleep apnea: A randomized control trial. Sleep, 34(4), 479–485.
https://pubmed.ncbi.nlm.nih.gov/21461326/ -
Lai, V., Tong, B. K., Tran, C., Ricciardiello, A., Donegan, M., Murray, N. P., Carberry, J. C., & Eckert, D. J. (2019). Combination therapy with mandibular advancement and expiratory positive airway pressure valves reduces obstructive sleep apnea severity. Sleep, 42(8), zsz119.
https://pubmed.ncbi.nlm.nih.gov/31180512/ -
Patel, A., & Kotecha, B. (2019). Minimally invasive radiofrequency surgery in sleep-disordered breathing. Healthcare (Basel, Switzerland), 7(3), 97.
https://pubmed.ncbi.nlm.nih.gov/31426606/ -
Camacho, M., Nesbitt, N. B., Lambert, E., Song, S. A., Chang, E. T., Liu, S. Y., Kushida, C. A., & Zaghi, S. (2017). Laser-assisted uvulopalatoplasty for obstructive sleep apnea: A systematic review and meta-analysis. Sleep, 40(3).
https://pubmed.ncbi.nlm.nih.gov/28201808/ -
Goh, Y. H., Abdullah, V., & Kim, S. W. (2019). Genioglossus advancement and hyoid surgery. Sleep Medicine Clinics, 14(1), 73–81.
https://pubmed.ncbi.nlm.nih.gov/30709536/ -
Tantawy, A. A., Askar, S. M., Amer, H. S., Awad, A., & El-Anwar, M. W. (2018). Hyoid bone suspension as a part of multilevel surgery for obstructive sleep apnea syndrome. International Archives of Otorhinolaryngology, 22(3), 266–270.
https://pubmed.ncbi.nlm.nih.gov/29983767/ -
Murphey, A. W., Kandl, J. A., Nguyen, S. A., Weber, A. C., & Gillespie, M. B. (2015). The effect of glossectomy for obstructive sleep apnea: A systematic review and meta-analysis. Otolaryngology–Head and Neck Surgery, 153(3), 334–342.
https://pubmed.ncbi.nlm.nih.gov/26183521/ -
Bielicki, P., Trojnar, A., Sobieraj, P., & Wasik, M. (2019). Smoking status in relation to obstructive sleep apnea severity (OSA) and cardiovascular comorbidity in patients with newly diagnosed OSA. Advances in Respiratory Medicine, 87(2), 103–109.
https://pubmed.ncbi.nlm.nih.gov/31038721/